News


Simple chromatographic method for detecting drugs of abuse in hair
A study presents a simplified LC-MS/MS method for detecting 26 drugs in hair using our MassTox® products. The method offers reduced sample preparation with reliable performance and is well suited for routine laboratory workflows and multiple drugs of abuse screening.


Ensuring accurate vitamin D measurement
Chromsystems participated in a global study to evaluate new vitamin D reference materials and was one of the few laboratories to meet all quality criteria. The results confirm the high quality of our products and the superiority of LC-MS/MS methods.
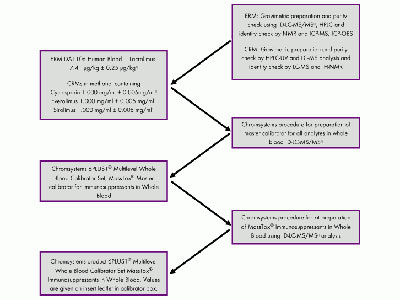
Study to establish more accurate cut-off values in newborn screening
A study shows that establishing specific and population-based cut-off values leads to a reduction in false-positive results.
Successful trade show appearance at Analytica
Our stand in Munich was a hub of activity, and a highlight was the live demonstration of MassSTAR, our automation solution for LC-MS/MS.
Automation in clinical LC-MS/MS
LC-MS-based testing offers accuracy, specificity, and a high speed of analysis, which is why it is being adopted across many clinical laboratories.
App Note: Therapeutic Drug Monitoring of Immunosuppressants with MassTox® Series A
Customers can use this protocol on a research-use basis, which demonstrates the analysis of more than 200 drugs plus immunosuppressants in the laboratory while also eliminating the need for changes to the chromatographic setup.
Study: Time to Rethink Amino Acid Analysis (AAA)
Quantitative AAA requires high sensitivity and specificity. Over the years, the gold standard technique has been Ion Exchange Chromatography (IEC). As with most techniques, IEC does have some drawbacks. A recent study by Carling et al suggests that it might be time to rethink quantitative amino acid analyses and compared IEC with LC-MS/MS as an alternative method.
Educational Webinar: Challenging the Status Quo of Amino Acid Analysis in Plasma
Dr. Rachel Carling, Scientific Director for Viapath, reports in this recorded webinar on a study that compares the current gold standard in quantitative amino acid analysis (AAA) - ion exchange chromatography (IEC) - with LC-MS/MS.
List: Metabolism Disorders in Amino Acid Profile
This table features exemplary amino acid metabolism disorders and associated changes in the amino acid profile that are covered by the MassChrom Amino Acid Analysis (AAA) assay.
Scientific Paper: Vitamin D Concentrations Are Lower in Patients with Positive PCR for SARS-CoV-2
A new study investigated the potential relationship between vitamin D blood concentrations and SARS-CoV-2 infections. Is the risk of an infection and the disease severity influenced by the vitamin D status? The scientists used the MassChrom® Vitamin D assay on a MassSTAR automation system.
Product Information Leaflets in Excel Format
Every package of calibrators and controls contains a product insert with necessary target values and calibrator concentrations. To make the data transfer more efficient, we are now providing Excel files of the inserts.
UDI Labelling of Our Products
We are currently integrating the new UDI code on our labels as part of the Unique Device Identification (UDI) System. UDIs are required only in the United States so far, but will become mandatory in Europe when the new IVDR comes into effect.
Article: Traceability in Clinical Diagnostics
The ultimate aim of traceability is to ensure that analytical results used for patient care are accurate as well as comparable over time and location. What we do to achieve that? Learn more in this article.
Article: TDM in ECMO Patients
During extracorporeal membrane oxygenation intensive care patients receive various antibiotics. If these drugs are not measured regularly, then patients are at risk of being underdosed and are unlikely to meet the MIC.
Article: Therapeutic Drug Monitoring in Intensive Care
This article discusses the importance of appropriate drug concentrations in antibiotic therapies to and help prevent over- and underdosing. This approach also reduces the risk of new multidrug resistant strains emerging and helps to provide the most effective drug dose to the patient.
TDM of clinically relevant antibiotics
Chromsystems offers a CE-IVD method for the quantitative determination of ampicillin, cefepime, ceftazidime, linezolid, meropenem and piperacillin in human serum and plasma samples. The robust HPLC method is simple to perform and easy to establish.
Article: Drugs of Abuse Testing with LC-MS/MS
In this article we compare LC-MS/MS with GC-MS currently considered as gold standard. The benefits of LC-MS/MS are demonstrated and it is shown how a 100% hydrolysis of glucuroniated drugs can be achieved.