Study: Time to Rethink Amino Acid Analysis (AAA)
The quantitative amino acid analysis (AAA) is crucial for the diagnosis of inherited diseases that affect amino acid metabolism. The most frequent are classical phenylketonuria (PKU) and Maple syrup urine disease (MSUD), but there are also many other rare diseases. Amino acid analysis requires high sensitivity and specificity. Over the years, the gold standard technique has been Ion Exchange Chromatography (IEC). As with most techniques, IEC does have some drawbacks, and a recent study by Carling et al suggests that it might be time to rethink how clinical laboratories approach quantitative amino acid analyses.


The Problems with Ion Exchange Chromatography (IEC)
Even though IEC is considered the go-to method in clinical diagnostic settings, its operation has been unchanged for over four decades. Nevertheless, there are enough drawbacks to warrant looking into other techniques: some of the practical disadvantages of IEC are a poor retention of acidic compounds, single point calibrations, and the need for post-experimental analysis programmes to negate some of these limitations. However, one major roadblock for clinical laboratories is a long analysis time of more than 120 minutes, which limits the speed of reporting results for clinically urgent samples and inhibits the efficiency in the routine laboratory workflow.
LC-MS/MS: A New Approach to AAA
A new study looked at LC-MS/MS as an alternative method. The researchers compared IEC with different commercial LC-MS/MS assays for the determination in human plasma samples, including the Chromsystems assay MassChrom® Amino Acid Analysis in Plasma/Serum. Generally, not all LC-MS/MS methods showed the performance required for all analytes. However, the analysis carried out with the MassChrom® assay showed a performance comparable to IEC, but with a higher degree of specificity and a much quicker analysis time of less than 19 minutes for 48 amino acids. “Sample preparation is quick and simple, taking approximately 20 minutes and utilising sensible volumes for a manual assay”, the authors said. The specificity was achieved via a combination of chromatographic resolution and selected reaction monitoring (SRM) approaches. “The underivatised approach has an impact on the sensitivity of the analytes with a small m/z.”, the authors also concluded. The Chromsystems assay was the only one in this study to also separate the isobaric compounds Leucine, Iso-Leucine and Allo-Leucine, eliminating the need for second line testing for these analytes.
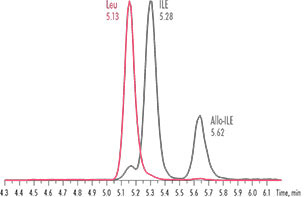

Fig.1: The Chromsystems-assay provides an efficient separation of all isobars such as Leu, Ile and Allo-Ile.
Conclusion
LC-MS/MS in general, and the MassChrom® assay in particular, demonstrated an analytical performance that is comparable to that of an IEC analysis. However, the amino acid analysis is performed much faster and with a higher degree of specificity. The availability of a commercial CE-IVD compliant reagent kit from Chromsystems can simplify reagent management procedures required for accreditation to UKAS standards. “The benefit of this in the routine clinical laboratory should not be underestimated”, the authors wrote.
The study ultimately concluded that because the same - or better - results can be obtain faster, then there is no longer a need to use IEC and it should no longer be classified as the gold standard method for plasma amino acid analysis. Results in the study were also presented in a webinar that you can watch on-demand.
Last Update 20th of April 2021
Dr Rachel Carling, Scientific Director for Viapath, reports on a study that compares the current gold standard in quantitative amino acid analysis (AAA) - ion exchange chromatography (IEC) with LC-MS/MS.


Dr. Rachel Carling, Consultant Scientist and Director of Biochemical Sciences, Viapath, UK
Rachel is working at Guy's and St Thomas’ Hospital where she is a Consultant Scientist, Director of Newborn Screening and Clinical Lead, Biochemical Sciences. She is also the Scientific Director for Viapath at the GSTT site. Her area of interest is inherited metabolic disease with a particular interest in tandem mass spectrometry.
Reference/Original Study:
Carling R. S. et al, Challenging the status quo: A comparison of ion exchange chromatography with liquid chromatography–mass spectrometry and liquid chromatography–tandem mass spectrometry methods for the measurement of amino acids in human plasma, Annals of Clinical Biochemistry¸ 2020, DOI: 10.1177/0004563220933303

