Parameter Set Antiepileptic Drugs - LC-MS/MS
Encompasses 26 analytes
3PLUS1® Multilevel Calibrator Set available
Part of the MassTox® TDM Series A
Method for automated sample preparation also available
CE-IVD validated product ready for IVDR within timeframes and transition periods specified by the IVDR 2017/746
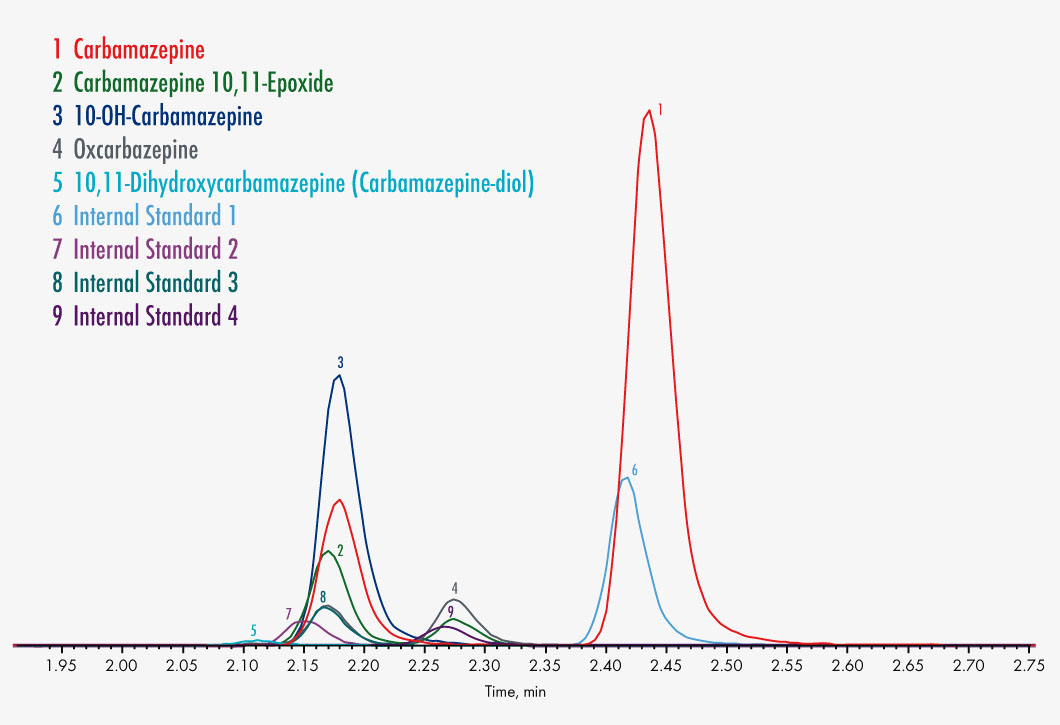





Carbamazepine
Carbamazepine-10,11-epoxide
10,11-Dihydroxycarbamazepine
10-OH-Carbamazepine
Ethosuximide
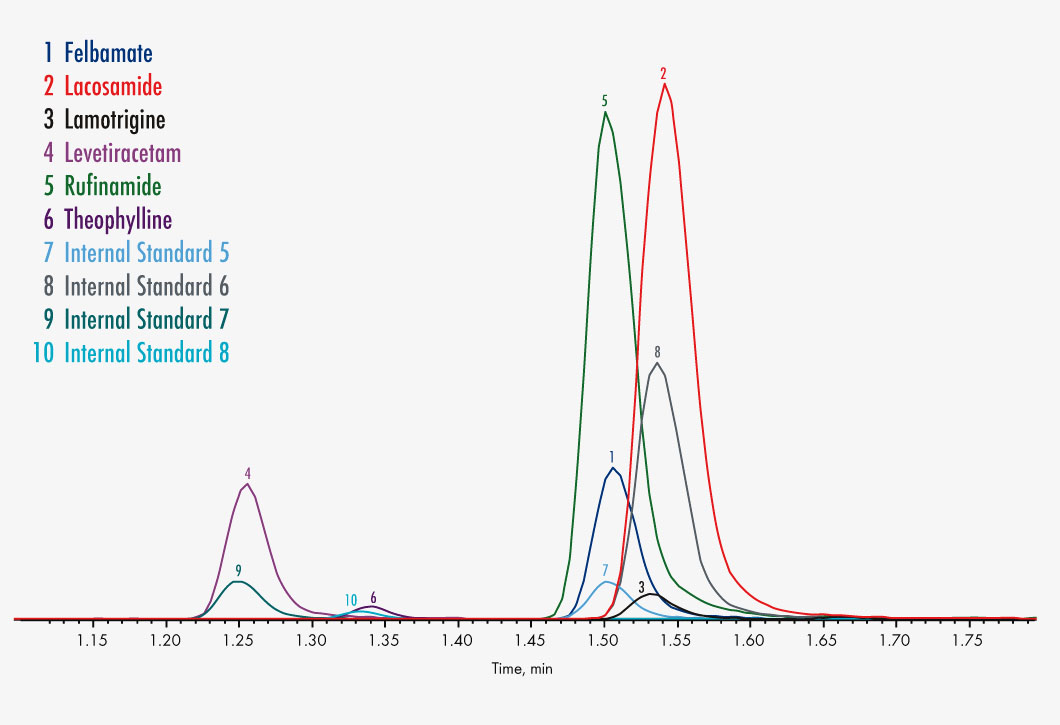
Felbamate
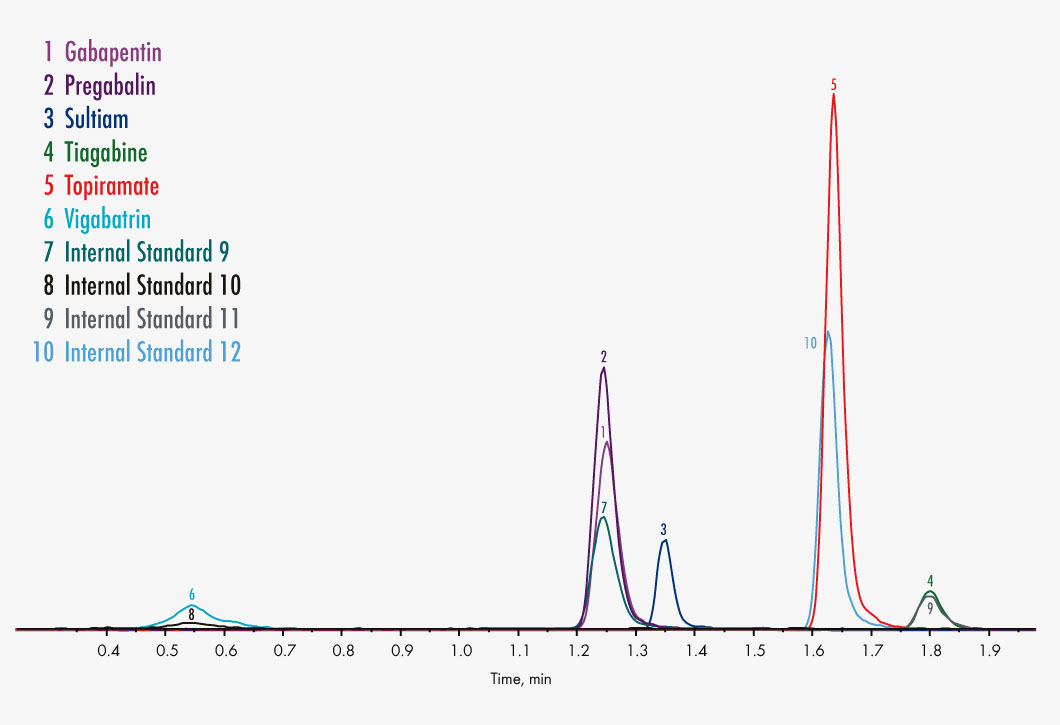
Gabapentin
Lacosamide
Lamotrigine
Levetiracetam (Keppra®)
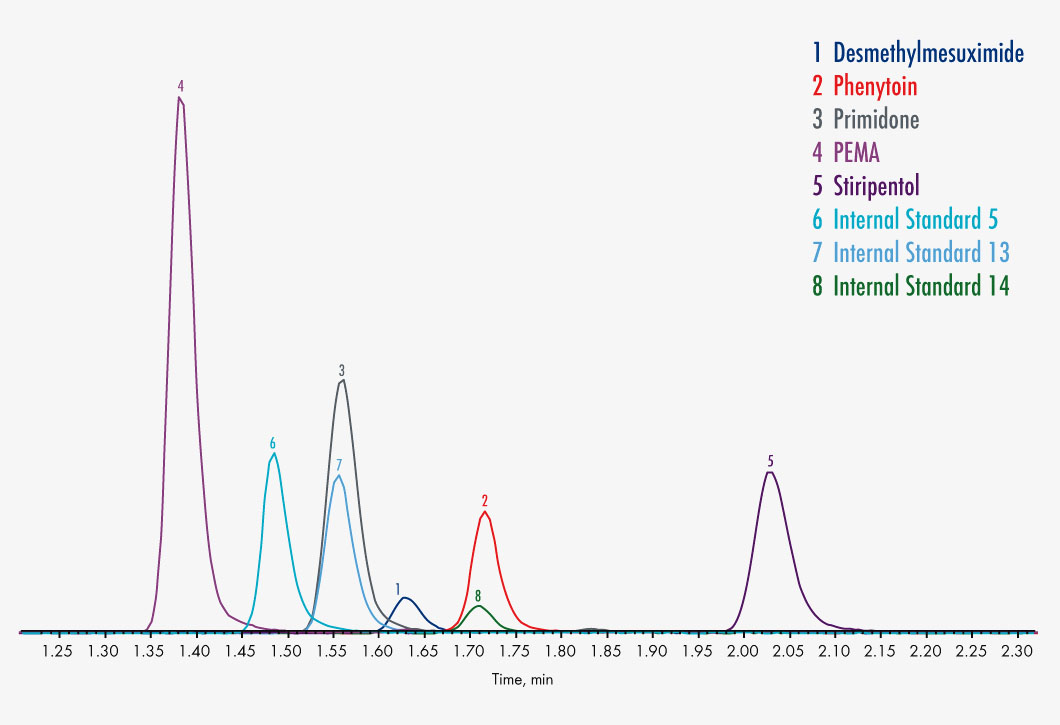
N-Desmethylmesuximide
Oxcarbazepine
Phenobarbital
Phenylethylmalonamide (PEMA)
Phenytoin
Pregabalin
Primidone
Rufinamide
Stiripentol
Sultiame
Theophylline
Tiagabine
Topiramate
Valproic acid
Vigabatrin
Zonisamid
Clinical relevance
Epileptic seizures are the result of synchronous discharges of neuron groups in the brain that can lead to sudden and involuntary stereotypical behavioural or sensory disorders. Numerous types of seizures have been described, each of which requires specialised therapy. The probability of them occurring depends on a number of factors. In addition to genetic predisposition, it is primarily exogenous factors that are relevant, such as accident, thrombosis, tumours or meningitis. Therapy with anticonvulsive medication (antiepileptics) leads to a reduction in seizures and sometimes even a complete elimination of seizures in most treated patients. The precondition for the antiepileptics to have a therapeutic effect is usually patient compliance, which means regular use of the medication. Thus, monitoring of the blood levels is essential, especially during initial dose setting.
MassTox® TDM Series A
The MassTox® TDM Series A is a modular system that enables the determination of 200 analytes without changing column or mobile phases, thereby minimising the workload in the laboratory.
It consists of 3 parts:
• MassTox® TDM Basic Kit A
• Specific MassTox® TDM Parameter Set (13 different parameter sets available)
• Analytical column MassTox® TDM MasterColumn® A
![]() More information about MassTox® TDM Series A
More information about MassTox® TDM Series A
| Method of Analysis | LC-MS/MS |
|---|---|
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | 0.1 – 7.0 mg/l |
| Upper Limit of Quantification | 1 – 300 mg/l |
| Intraassay | CV = 2 – 8 % |
| Interassay | CV = 4 – 9 % |
| Recovery | 84 – 115 % |
| Specimen | Serum/Plasma |
| Sample Preparation |
|
| Run Time | 2.5 – 3.5 min |
| Injection Volume | 0.2 – 50 µl |
| Gradient | Starting point: 0% Mobile Phase 2 Group 1 Group 2 and 3 Group 4 and 5 |
| Ionisation | ESI positive and negative |
| MS/MS Mode | MRM |
| Additional Info | We recommend to set the scan time to a value that allows to achieve a minimum of 10 data points over the whole peak width. |
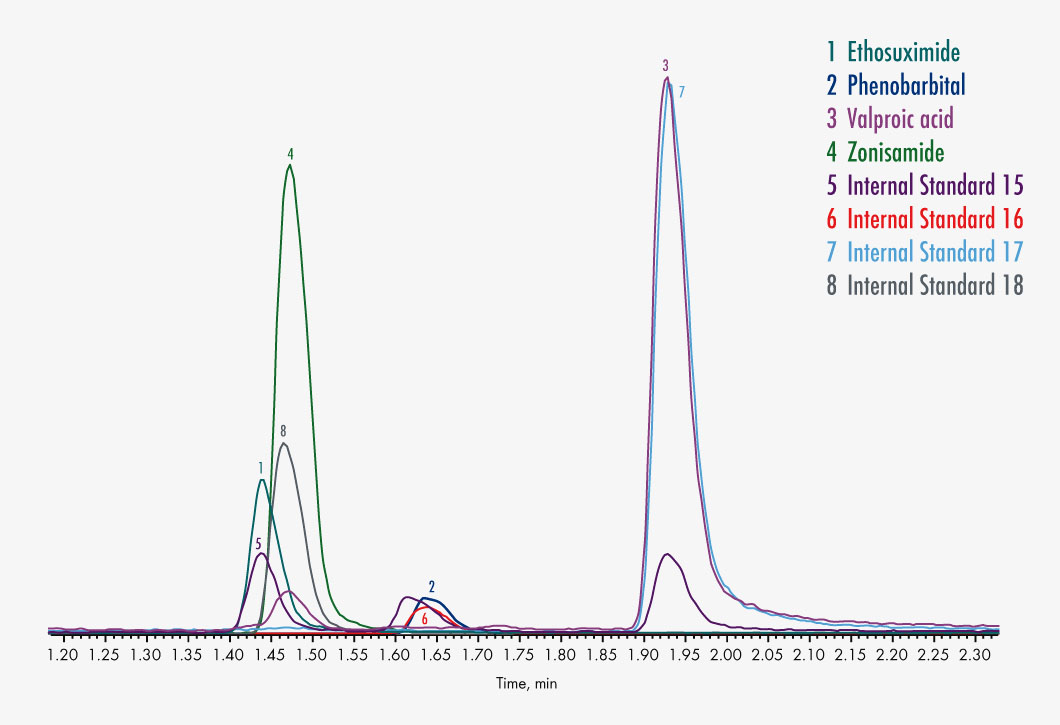
| Parameters | 10-OH-Carbamazepine, Carbamazepine, Ethosuximide, Felbamate, Gabapentin, Lacosamide, Lamotrigine, N-Desmethylmesuximide, Phenobarbital, Phenytoin, Pregabalin, Primidone, Rufinamide, Stiripentol, Sultiame, Theophylline, Tiagabine, Topiramate, Valproic Acid, Vigabatrin, Zonisamide |
-
 3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS -
 MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XT
MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
-
 Internal Standard Mix Antiepileptic DrugsOrder No.: 92546Component of the Parameter Set Antiepileptic Drugs, available separately
Internal Standard Mix Antiepileptic DrugsOrder No.: 92546Component of the Parameter Set Antiepileptic Drugs, available separately
-
 MassTox® TDM MasterColumn® AOrder No.: 92110
MassTox® TDM MasterColumn® AOrder No.: 92110Analytical column for MassTox® TDM Series A - LC-MS/MS
-
 Tuning Mix Antiepileptic Drugs/EXTENDED 1Order No.: 92034/XTTuning Mix 1 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 1Order No.: 92034/XTTuning Mix 1 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 2Order No.: 92035/XTTuning Mix 2 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 2Order No.: 92035/XTTuning Mix 2 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 3Order No.: 92036/XTTuning Mix 3 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 3Order No.: 92036/XTTuning Mix 3 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 4Order No.: 92037/XTTuning Mix 4 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 4Order No.: 92037/XTTuning Mix 4 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 5Order No.: 92038/XTTuning Mix 5 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 5Order No.: 92038/XTTuning Mix 5 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Basic Kit A for 200 Tests - LC-MS/MSOrder No.: 92111/200
Basic Kit A for 200 Tests - LC-MS/MSOrder No.: 92111/200Part of the MassTox® TDM Series A
Modular system for therapeutic drug monitoring
Provides all components required for sample prep and all mobile phasesValidated according to IVDR
-
 Basic Kit A for 1000 Tests - LC-MS/MSOrder No.: 92111/1000
Basic Kit A for 1000 Tests - LC-MS/MSOrder No.: 92111/1000Part of the MassTox® TDM Series A
Modular system for therapeutic drug monitoring
Provides all components required for sample prep and all mobile phasesValidated according to IVDR
-
 Basic Kit A for 1000 Tests with 96 Well Filter Plates - LC-MS/MSOrder No.: 92111/1000/F
Basic Kit A for 1000 Tests with 96 Well Filter Plates - LC-MS/MSOrder No.: 92111/1000/FPart of the MassTox® TDM Series A
Modular system for therapeutic drug monitoring
Provides all components required for sample prep and all mobile phases
Sample preparation with 96 Well Filter PlatesValidated according to IVDR
-
 3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
-
 MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XT
MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS










Carbamazepine
Carbamazepine-10,11-epoxide
10,11-Dihydroxycarbamazepine
10-OH-Carbamazepine
Ethosuximide
Felbamate
Gabapentin
Lacosamide
Lamotrigine
Levetiracetam (Keppra®)
N-Desmethylmesuximide
Oxcarbazepine
Phenobarbital
Phenylethylmalonamide (PEMA)
Phenytoin
Pregabalin
Primidone
Rufinamide
Stiripentol
Sultiame
Theophylline
Tiagabine
Topiramate
Valproic acid
Vigabatrin
Zonisamid
Clinical relevance
Epileptic seizures are the result of synchronous discharges of neuron groups in the brain that can lead to sudden and involuntary stereotypical behavioural or sensory disorders. Numerous types of seizures have been described, each of which requires specialised therapy. The probability of them occurring depends on a number of factors. In addition to genetic predisposition, it is primarily exogenous factors that are relevant, such as accident, thrombosis, tumours or meningitis. Therapy with anticonvulsive medication (antiepileptics) leads to a reduction in seizures and sometimes even a complete elimination of seizures in most treated patients. The precondition for the antiepileptics to have a therapeutic effect is usually patient compliance, which means regular use of the medication. Thus, monitoring of the blood levels is essential, especially during initial dose setting.
MassTox® TDM Series A
The MassTox® TDM Series A is a modular system that enables the determination of 200 analytes without changing column or mobile phases, thereby minimising the workload in the laboratory.
It consists of 3 parts:
• MassTox® TDM Basic Kit A
• Specific MassTox® TDM Parameter Set (13 different parameter sets available)
• Analytical column MassTox® TDM MasterColumn® A
![]() More information about MassTox® TDM Series A
More information about MassTox® TDM Series A
| Method of Analysis | LC-MS/MS |
|---|---|
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | 0.1 – 7.0 mg/l |
| Upper Limit of Quantification | 1 – 300 mg/l |
| Intraassay | CV = 2 – 8 % |
| Interassay | CV = 4 – 9 % |
| Recovery | 84 – 115 % |
| Specimen | Serum/Plasma |
| Sample Preparation |
|
| Run Time | 2.5 – 3.5 min |
| Injection Volume | 0.2 – 50 µl |
| Gradient | Starting point: 0% Mobile Phase 2 Group 1 Group 2 and 3 Group 4 and 5 |
| Ionisation | ESI positive and negative |
| MS/MS Mode | MRM |
| Additional Info | We recommend to set the scan time to a value that allows to achieve a minimum of 10 data points over the whole peak width. |
| Parameters | 10-OH-Carbamazepine, Carbamazepine, Ethosuximide, Felbamate, Gabapentin, Lacosamide, Lamotrigine, N-Desmethylmesuximide, Phenobarbital, Phenytoin, Pregabalin, Primidone, Rufinamide, Stiripentol, Sultiame, Theophylline, Tiagabine, Topiramate, Valproic Acid, Vigabatrin, Zonisamide |
-
 3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS -
 MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XT
MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
-
 Internal Standard Mix Antiepileptic DrugsOrder No.: 92546Component of the Parameter Set Antiepileptic Drugs, available separately
Internal Standard Mix Antiepileptic DrugsOrder No.: 92546Component of the Parameter Set Antiepileptic Drugs, available separately
-
 MassTox® TDM MasterColumn® AOrder No.: 92110
MassTox® TDM MasterColumn® AOrder No.: 92110Analytical column for MassTox® TDM Series A - LC-MS/MS
-
 Tuning Mix Antiepileptic Drugs/EXTENDED 1Order No.: 92034/XTTuning Mix 1 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 1Order No.: 92034/XTTuning Mix 1 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 2Order No.: 92035/XTTuning Mix 2 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 2Order No.: 92035/XTTuning Mix 2 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 3Order No.: 92036/XTTuning Mix 3 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 3Order No.: 92036/XTTuning Mix 3 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 4Order No.: 92037/XTTuning Mix 4 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 4Order No.: 92037/XTTuning Mix 4 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Tuning Mix Antiepileptic Drugs/EXTENDED 5Order No.: 92038/XTTuning Mix 5 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS
Tuning Mix Antiepileptic Drugs/EXTENDED 5Order No.: 92038/XTTuning Mix 5 for the Parameter Sets Antiepileptic Drugs - LC-MS/MS -
 Basic Kit A for 200 Tests - LC-MS/MSOrder No.: 92111/200
Basic Kit A for 200 Tests - LC-MS/MSOrder No.: 92111/200Part of the MassTox® TDM Series A
Modular system for therapeutic drug monitoring
Provides all components required for sample prep and all mobile phasesValidated according to IVDR
-
 Basic Kit A for 1000 Tests - LC-MS/MSOrder No.: 92111/1000
Basic Kit A for 1000 Tests - LC-MS/MSOrder No.: 92111/1000Part of the MassTox® TDM Series A
Modular system for therapeutic drug monitoring
Provides all components required for sample prep and all mobile phasesValidated according to IVDR
-
 Basic Kit A for 1000 Tests with 96 Well Filter Plates - LC-MS/MSOrder No.: 92111/1000/F
Basic Kit A for 1000 Tests with 96 Well Filter Plates - LC-MS/MSOrder No.: 92111/1000/FPart of the MassTox® TDM Series A
Modular system for therapeutic drug monitoring
Provides all components required for sample prep and all mobile phases
Sample preparation with 96 Well Filter PlatesValidated according to IVDR
-
 3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
3PLUS1® Multilevel Plasma Calibrator Set Antiepileptic Drugs/EXTENDEDOrder No.: 92025/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS
-
 MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XT
MassCheck® Antiepileptic Drugs/EXTENDED Plasma ControlsOrder No.: 0249/XT; 0250/XT; 0251/XTMassTox® TDM Series A Antiepileptic Drugs in Serum/Plasma – LC-MS/MS






