Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
No interference with concomitant drug therapy
Identical sample preparation to reagent kit order no. 22000
Option of combined analysis with 22000/F
CE-IVD validated product ready for IVDR within timeframes and transition periods specified by the IVDR 2017/746
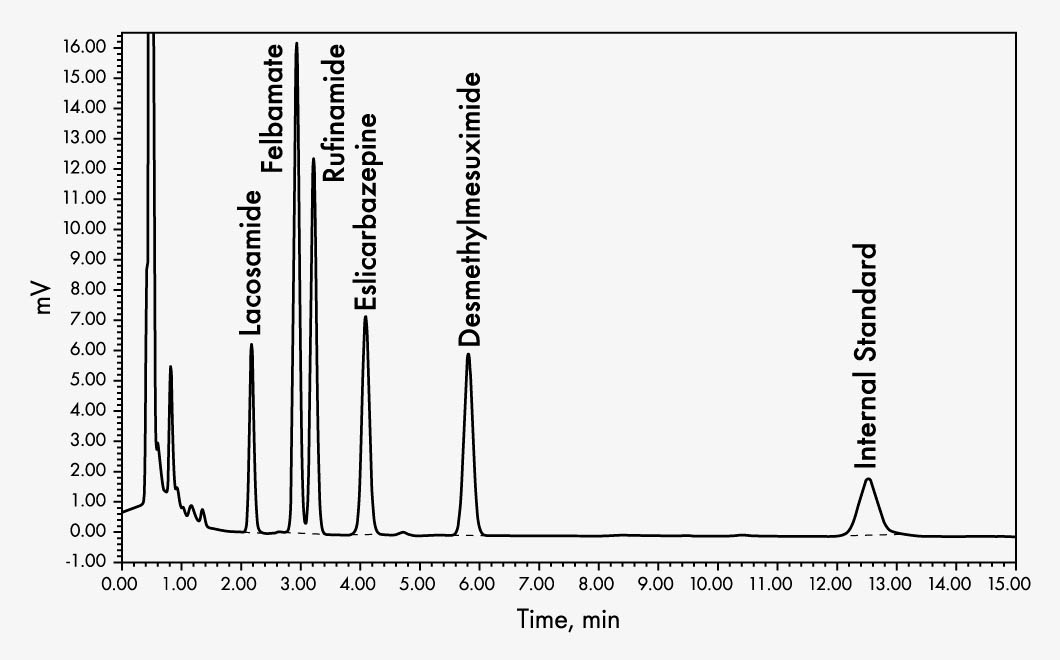

Eslicarbazepine
Felbamate
Lacosamide
N-Desmethylmesuximide
Rufinamide
Clinical relevance
Rufinamide, felbamate and mesuximide are approved in the EU for concomitant therapy in combination with other antiepileptic drugs (AEDs). They are primarily used to treat therapy-resistant epilepsies and Lennox–Gastaut syndrome, and are also suitable for difficult to treat epilepsies in children. Rufinamide and felbamate influence the effectiveness of other AEDs and contraceptive products. Eslicarbazepine acetate and lacosamide are also used in combination with other AEDs to treat partial epileptic seizures.
The blood levels of these medications must be determined to maintain the therapeutic range due to their pharmacodynamics and toxicities. That also includes measuring the levels of the metabolites N-desmethyl mesuximide and eslicarbazepine.
Product advantages
- Undisturbed analysis for comedication with other antiepileptics
- Sample preparation is identical to the reagent kit "Antiepileptics for HPLC", # 22000
- Combined analysis with 22000/F is possible
Enhanced HPLC analysis of the antiepileptics enables the reliable determination of the AEDs rufinamide, felbamate, lacosamide, eslicarbazepine and N-desmethyl mesuximide. Sample preparation requires only one precipitation step and is identical to that of the Chromsystems kit order no. 22000. It has been tested for interference from other AEDs and can therefore guarantee undisturbed analysis even in the case of concomitant therapy with other AEDs.
Our TDM Parameter Set Antiepileptic Drugs from the MassTox® TDM Series A is available as an alternative for the measurement of these analytes with LC-MS/MS.
| Method of Analysis | HPLC |
|---|---|
| Number of Tests | 100 |
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | 0.5–1.0 mg/l |
| Upper Limit of Quantification | up to 200 mg/l |
| Intraassay | CV < 2 % |
| Interassay | CV < 2 % |
| Recovery | 100 % |
| Specimen | Serum/Plasma |
| Sample Preparation |
|
| Run Time | < 15 min |
| Injection Volume | 20 µl |
| Flow Rate | 2.5 ml/min |
| Column Temperature | ambient temperature (~25 °C) |
| Gradient | isokratic |
| Wavelengths | 204 nm |
| Additional Info | Isocratic HPLC system with UV detector. |
| Parameters | Eslicarbazepine, Felbamate, Lacosamide, N-Desmethylmesuximide, Rufinamide |
-
 Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
-
 HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC
HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC
-
 Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
-
 HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC
HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC


Eslicarbazepine
Felbamate
Lacosamide
N-Desmethylmesuximide
Rufinamide
Clinical relevance
Rufinamide, felbamate and mesuximide are approved in the EU for concomitant therapy in combination with other antiepileptic drugs (AEDs). They are primarily used to treat therapy-resistant epilepsies and Lennox–Gastaut syndrome, and are also suitable for difficult to treat epilepsies in children. Rufinamide and felbamate influence the effectiveness of other AEDs and contraceptive products. Eslicarbazepine acetate and lacosamide are also used in combination with other AEDs to treat partial epileptic seizures.
The blood levels of these medications must be determined to maintain the therapeutic range due to their pharmacodynamics and toxicities. That also includes measuring the levels of the metabolites N-desmethyl mesuximide and eslicarbazepine.
Product advantages
- Undisturbed analysis for comedication with other antiepileptics
- Sample preparation is identical to the reagent kit "Antiepileptics for HPLC", # 22000
- Combined analysis with 22000/F is possible
Enhanced HPLC analysis of the antiepileptics enables the reliable determination of the AEDs rufinamide, felbamate, lacosamide, eslicarbazepine and N-desmethyl mesuximide. Sample preparation requires only one precipitation step and is identical to that of the Chromsystems kit order no. 22000. It has been tested for interference from other AEDs and can therefore guarantee undisturbed analysis even in the case of concomitant therapy with other AEDs.
Our TDM Parameter Set Antiepileptic Drugs from the MassTox® TDM Series A is available as an alternative for the measurement of these analytes with LC-MS/MS.
| Method of Analysis | HPLC |
|---|---|
| Number of Tests | 100 |
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | 0.5–1.0 mg/l |
| Upper Limit of Quantification | up to 200 mg/l |
| Intraassay | CV < 2 % |
| Interassay | CV < 2 % |
| Recovery | 100 % |
| Specimen | Serum/Plasma |
| Sample Preparation |
|
| Run Time | < 15 min |
| Injection Volume | 20 µl |
| Flow Rate | 2.5 ml/min |
| Column Temperature | ambient temperature (~25 °C) |
| Gradient | isokratic |
| Wavelengths | 204 nm |
| Additional Info | Isocratic HPLC system with UV detector. |
| Parameters | Eslicarbazepine, Felbamate, Lacosamide, N-Desmethylmesuximide, Rufinamide |
-
 Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
-
 HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC
HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC
-
 Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
Rufinamide, Felbamate, Lacosamide Serum Calibration StandardOrder no.: 21003Rufinamide, Felbamate and Lacosamide in Serum/Plasma - HPLC
-
 HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC
HPLC Column Antiepileptics in Serum/Plasma - Fast ElutionOrder no.: 22100/FAntiepileptics in Serum/Plasma - HPLC