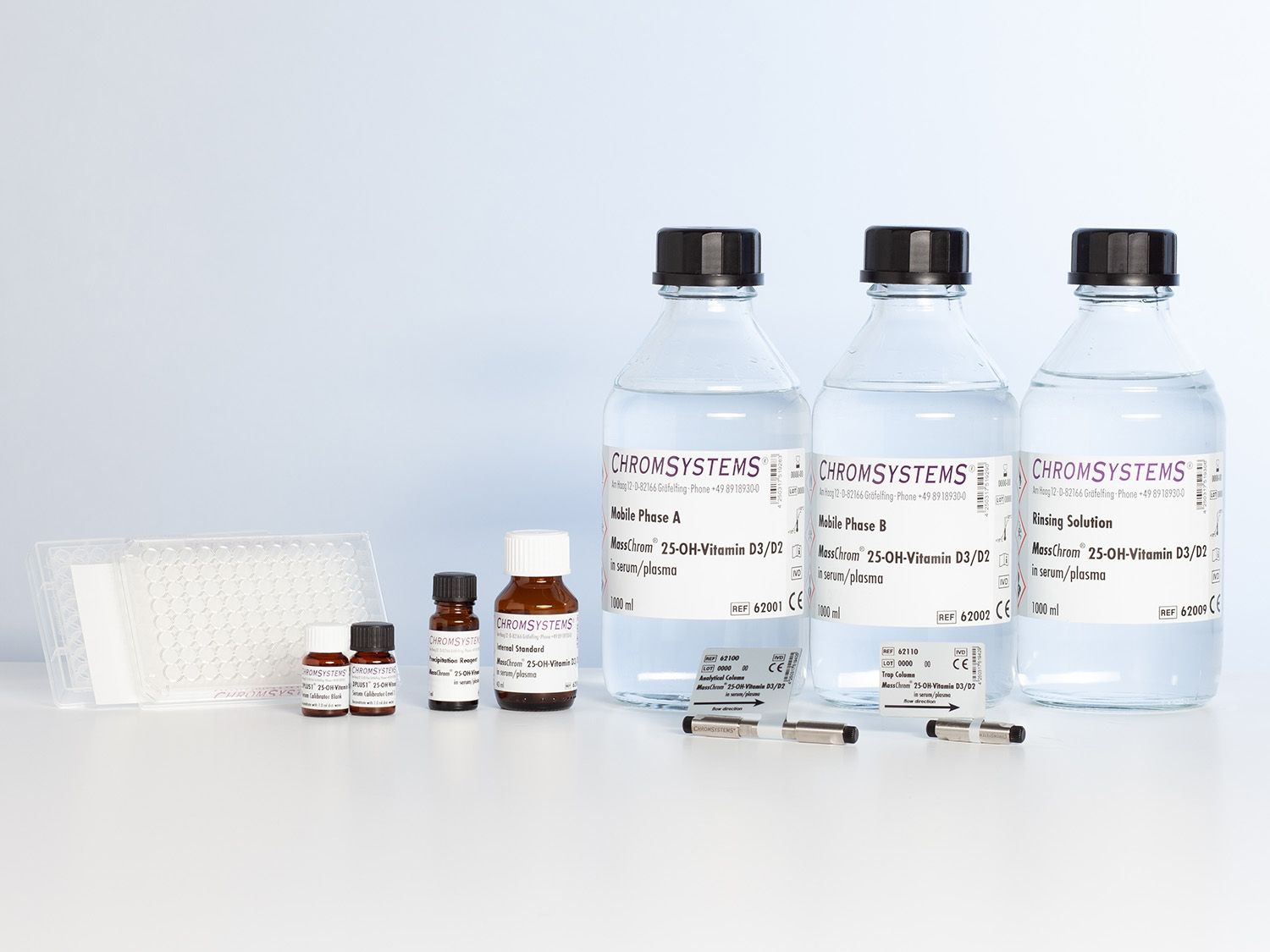
MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
For sample preparation with 96 Well Filter Plates
Assay validated according to IVDR (=> Declaration of Conformity)
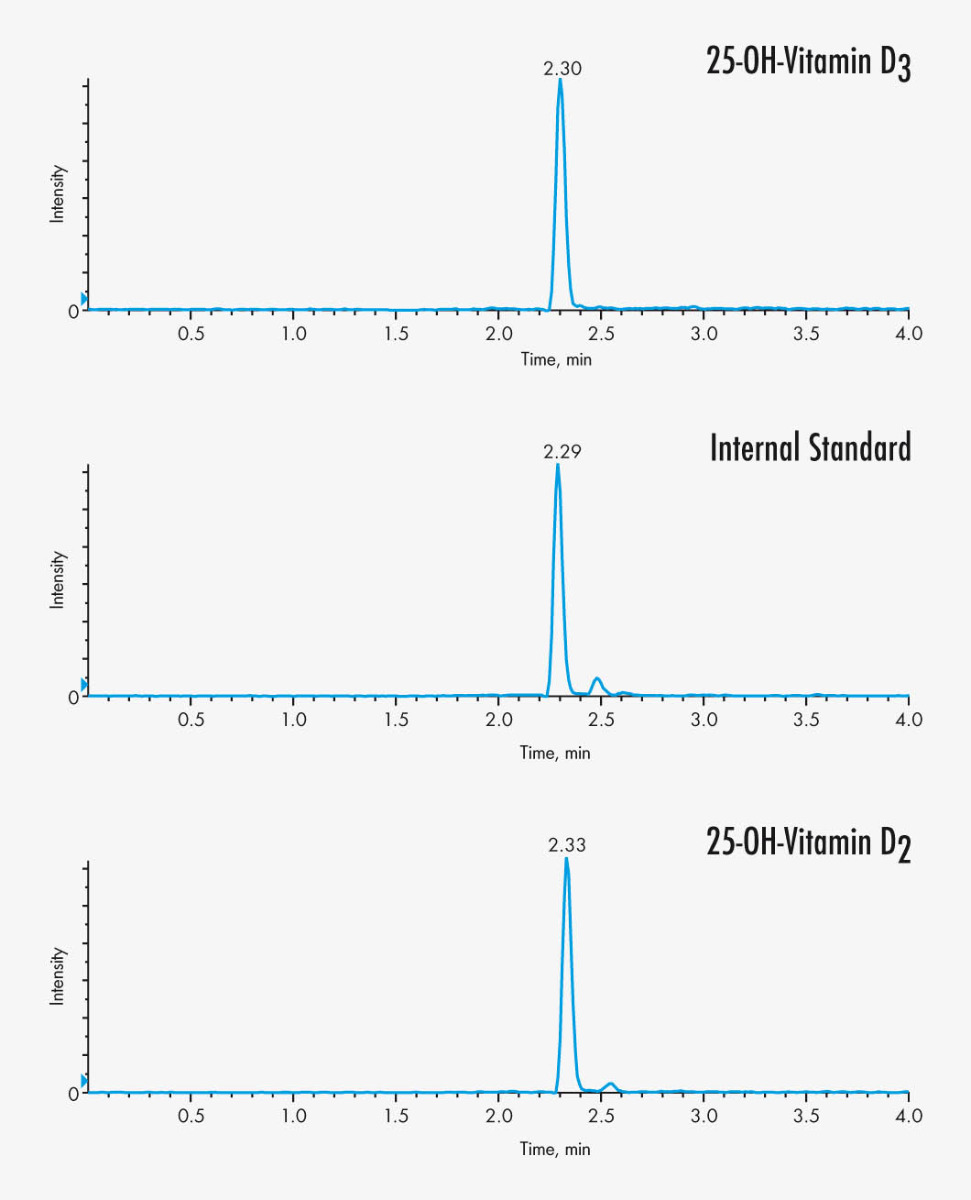

25-OH-Vitamin D2
25-OH-Vitamin D3
MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma by LC-MS/MS
Kit for 1000 tests, including Analytical Column, Trap Column, 3PLUS1® Multilevel Calibrator Set and MassCheck® Controls
For sample preparation with 96 Well Filter Plates
Clinical relevance
This assay allows the quantitative determination of 25-hydroxycholecalciferol (25-OH-vitamin D3) and 25-hydroxyergocalciferol (25-OH-vitamin D2) in human plasma or serum samples.
It is intended to be used for screening and/or monitoring of 25-OH-vitamin D3/D2 levels where indicated:
- in patients with suspected vitamin D deficiency,
- in patients with suspected vitamin D insufficiency, or
- in patients with suspected vitamin D excess, and/or
- in patients under vitamin D supplementation therapy.
The test kit is further intended to be used as an aid to diagnosis of diseases for which determination of vitamin D levels is indicated.
Assay characteristics
Manual sample preparation is limited to simple and effective protein precipitation. A trap column concentrates the analytes and separates interfering substances. A two-position-six-port-valve connects the trap column to an HPLC column where the chromatographic separation takes place. The use of atmospheric pressure chemical ionisation (APCI) and a deuterated internal standard assures precision and robustness and minimises ion suppression effects.
This kit does not separate 3-epi-25-OH-vitamin D3 and 3-epi-25-OH-vitamin D2 from 25-OH-vitamin D3 and 25-OH-vitamin D2, respectively, and should therefore only be used to analyse samples that contain none or just negligible amounts of these metabolites.
We recommend the use of the assay MassChrom® 25-OH-Vitamin D3/D2 and 3-epi-25-OH-Vitamin D3 in Serum/Plasma (order no. 62062) for chromatographic separation of the diastereomers and quantitation of 3-epi-25-OH-vitamin D3 to ensure correct assessment of the vitamin D status in patient samples that might contain 3-epi-25-OH-vitamin D3/D2.
Detailed performance evaluation data for this assay can be found in Appendices II and III of the instruction manual.
| Method of Analysis | LC-MS/MS |
|---|---|
| Number of Tests | 1000 |
| Please note | The information listed here, including the sample preparation, is not sufficient for using the product. Please read the information provided in the instruction manual, which includes detailed information on limitations associated with the use of the product in line with its intended purpose. Detailed performance evaluation data for this assay can be found in Appendices II and III of the instruction manual. |
| Lower and Upper Limit of Quantitation | 25-OH-Vitamin D3: 1.0 µg/l – 250 µg/l Different systems might show different performance data. |
| Specimen | Serum/Plasma |
| Sample Preparation | The information on the sample preparation presented here is not sufficient for use in the laboratory. For a detailed step by step description, please refer to the instruction manual. Sample preparation with 96 well filter plates:
|
| Run Time | 5.0 or 6.0 min (depending on gradient system) |
| Injection Volume | ≤ 50 µl (mass spectrometer dependent) |
| Column Temperature | Analytical Column and Trap Column: +20 to +25 °C |
| Ionisation | APCI positive |
| MS/MS Mode | MRM |
| Parameters | 25-OH-Vitamin D2, 25-OH-Vitamin D3 |
-
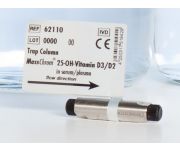
 Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100
Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
-
 3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS -
 MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256
MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC/MS-MS
-
 Tuning Mix 25-OH-Vitamin D3/D2Order No.: 62015
Tuning Mix 25-OH-Vitamin D3/D2Order No.: 62015MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
-
 3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
-
 MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256
MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC/MS-MS
-
 Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100
Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS


25-OH-Vitamin D2
25-OH-Vitamin D3
MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma by LC-MS/MS
Kit for 1000 tests, including Analytical Column, Trap Column, 3PLUS1® Multilevel Calibrator Set and MassCheck® Controls
For sample preparation with 96 Well Filter Plates
Clinical relevance
This assay allows the quantitative determination of 25-hydroxycholecalciferol (25-OH-vitamin D3) and 25-hydroxyergocalciferol (25-OH-vitamin D2) in human plasma or serum samples.
It is intended to be used for screening and/or monitoring of 25-OH-vitamin D3/D2 levels where indicated:
- in patients with suspected vitamin D deficiency,
- in patients with suspected vitamin D insufficiency, or
- in patients with suspected vitamin D excess, and/or
- in patients under vitamin D supplementation therapy.
The test kit is further intended to be used as an aid to diagnosis of diseases for which determination of vitamin D levels is indicated.
Assay characteristics
Manual sample preparation is limited to simple and effective protein precipitation. A trap column concentrates the analytes and separates interfering substances. A two-position-six-port-valve connects the trap column to an HPLC column where the chromatographic separation takes place. The use of atmospheric pressure chemical ionisation (APCI) and a deuterated internal standard assures precision and robustness and minimises ion suppression effects.
This kit does not separate 3-epi-25-OH-vitamin D3 and 3-epi-25-OH-vitamin D2 from 25-OH-vitamin D3 and 25-OH-vitamin D2, respectively, and should therefore only be used to analyse samples that contain none or just negligible amounts of these metabolites.
We recommend the use of the assay MassChrom® 25-OH-Vitamin D3/D2 and 3-epi-25-OH-Vitamin D3 in Serum/Plasma (order no. 62062) for chromatographic separation of the diastereomers and quantitation of 3-epi-25-OH-vitamin D3 to ensure correct assessment of the vitamin D status in patient samples that might contain 3-epi-25-OH-vitamin D3/D2.
Detailed performance evaluation data for this assay can be found in Appendices II and III of the instruction manual.
| Method of Analysis | LC-MS/MS |
|---|---|
| Number of Tests | 1000 |
| Please note | The information listed here, including the sample preparation, is not sufficient for using the product. Please read the information provided in the instruction manual, which includes detailed information on limitations associated with the use of the product in line with its intended purpose. Detailed performance evaluation data for this assay can be found in Appendices II and III of the instruction manual. |
| Lower and Upper Limit of Quantitation | 25-OH-Vitamin D3: 1.0 µg/l – 250 µg/l Different systems might show different performance data. |
| Specimen | Serum/Plasma |
| Sample Preparation | The information on the sample preparation presented here is not sufficient for use in the laboratory. For a detailed step by step description, please refer to the instruction manual. Sample preparation with 96 well filter plates:
|
| Run Time | 5.0 or 6.0 min (depending on gradient system) |
| Injection Volume | ≤ 50 µl (mass spectrometer dependent) |
| Column Temperature | Analytical Column and Trap Column: +20 to +25 °C |
| Ionisation | APCI positive |
| MS/MS Mode | MRM |
| Parameters | 25-OH-Vitamin D2, 25-OH-Vitamin D3 |
-
 Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100
Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
-
 3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS -
 MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256
MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC/MS-MS
-
 Tuning Mix 25-OH-Vitamin D3/D2Order No.: 62015
Tuning Mix 25-OH-Vitamin D3/D2Order No.: 62015MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
-
 3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
3PLUS1® Multilevel Serum Calibrator Set 25-OH-Vitamin D3/D2Order No.: 62028MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS
-
 MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256
MassCheck® 25-OH-Vitamin D3/D2 Serum ControlsOrder No.: 0221/0222/0223/0256MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC/MS-MS
-
 Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100
Analytical Column 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MSOrder No.: 62100MassChrom® 25-OH-Vitamin D3/D2 in Serum/Plasma - LC-MS/MS