Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Low limit of quantification
Robust UV-VIS detection
Metabolite included
CE-IVD validated product ready for IVDR within timeframes and transition periods specified by the IVDR 2017/746
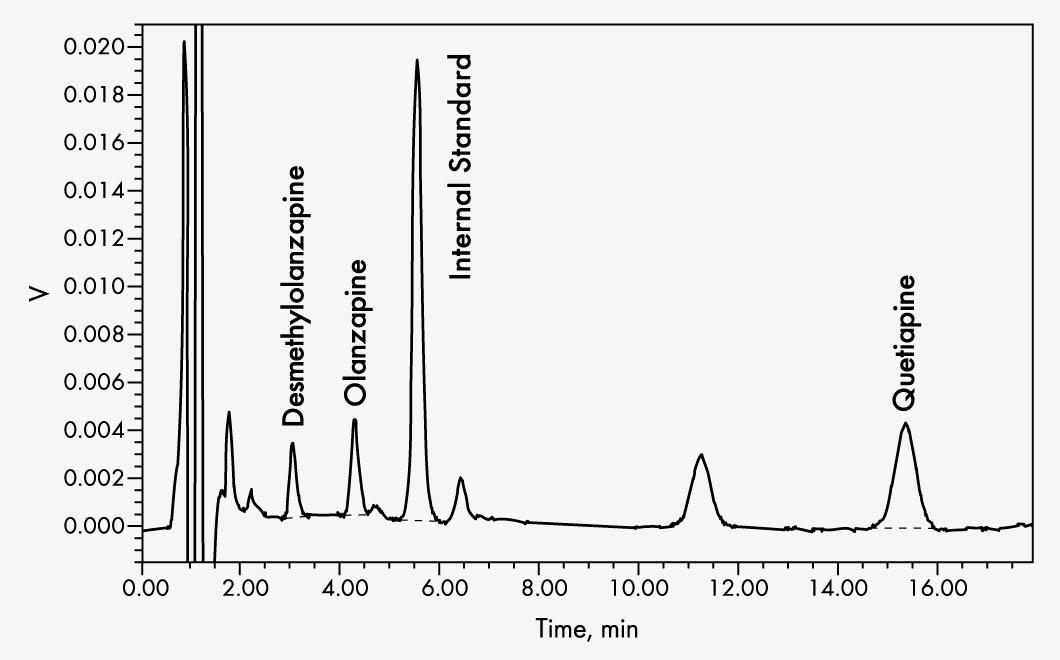

Olanzapine
N-Desmethylolanzapine
Quetiapine
Clinical relevance
Olanzapine and quetiapine are highly effective antipsychotic agents used to treat schizophrenic psychosis. Like clozapine, they are both atypical neuroleptics which are characterised by low levels of side effects in the extrapyramidal motor system (dyskinesia, Parkinson’s disease). Quetiapine is structurally related to olanzapine and is approved as a neuroleptic, but it shows a different receptor profile.
It is essential that the serum levels of these psychotropic drugs be monitored to ensure an optimal therapeutic effect and to minimise any undesirable side effects resulting from overdose. Furthermore, patient compliance gets checked and confirmed by this procedure.
Product advantages
- Low limit of quantification
- Robust UV-VIS detection
- Metabolite analysis included
This assay can be used to reliably determine the levels of olanzapine, its metabolite desmethylolanzapine, and quetiapine in serum or plasma. The sample preparation is based on an efficient and selective solid phase extraction including an internal standard. The chromatographic separation is run on an isocratic HPLC system with UV-VIS detector. When used with additional specific calibrators and controls, perazine and citalopram can also be determined in serum or plasma with this kit.
Our TDM Parameter Set Neuroleptics 1/XT from the MassTox® TDM Series A is available as an alternative for the measurement of these analytes using LC-MS/MS.
| Method of Analysis | HPLC |
|---|---|
| Number of Tests | 100 |
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | Olanzapine/Desmethylolanzapine: approx. 3.5 ng/ml Quetiapine: approx. 11 ng/ml |
| Upper Limit of Quantification | Olanzapine/Desmethylolanzapine: up to 1000 ng/ml |
| Intraassay | Olanzapine/Desmethylolanzapine: CV < 4.8 % |
| Interassay | Olanzapine/Desmethylolanzapine: CV < 9.6 % |
| Recovery | Olanzapine/Desmethylolanzapine: 95 %; Quetiapine: 96 % |
| Specimen | Serum/Plasma |
| Sample Preparation | Extraction
|
| Run Time | 18 min |
| Injection Volume | 50 µl |
| Flow Rate | 0.6 ml/min |
| Column Temperature | ambient (~ 25 °C) |
| Gradient | isocratic |
| Wavelengths | 254 nm |
| Additional Info | Any isocratic HLPC system with UV-VIS detector is suitable. |
| Parameters | N-Desmethylolanzapine, Olanzapine, Quetiapine |
-
 Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
-
 HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC -
 Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
-
 Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC -
 Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
-
 HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC


Olanzapine
N-Desmethylolanzapine
Quetiapine
Clinical relevance
Olanzapine and quetiapine are highly effective antipsychotic agents used to treat schizophrenic psychosis. Like clozapine, they are both atypical neuroleptics which are characterised by low levels of side effects in the extrapyramidal motor system (dyskinesia, Parkinson’s disease). Quetiapine is structurally related to olanzapine and is approved as a neuroleptic, but it shows a different receptor profile.
It is essential that the serum levels of these psychotropic drugs be monitored to ensure an optimal therapeutic effect and to minimise any undesirable side effects resulting from overdose. Furthermore, patient compliance gets checked and confirmed by this procedure.
Product advantages
- Low limit of quantification
- Robust UV-VIS detection
- Metabolite analysis included
This assay can be used to reliably determine the levels of olanzapine, its metabolite desmethylolanzapine, and quetiapine in serum or plasma. The sample preparation is based on an efficient and selective solid phase extraction including an internal standard. The chromatographic separation is run on an isocratic HPLC system with UV-VIS detector. When used with additional specific calibrators and controls, perazine and citalopram can also be determined in serum or plasma with this kit.
Our TDM Parameter Set Neuroleptics 1/XT from the MassTox® TDM Series A is available as an alternative for the measurement of these analytes using LC-MS/MS.
| Method of Analysis | HPLC |
|---|---|
| Number of Tests | 100 |
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | Olanzapine/Desmethylolanzapine: approx. 3.5 ng/ml Quetiapine: approx. 11 ng/ml |
| Upper Limit of Quantification | Olanzapine/Desmethylolanzapine: up to 1000 ng/ml |
| Intraassay | Olanzapine/Desmethylolanzapine: CV < 4.8 % |
| Interassay | Olanzapine/Desmethylolanzapine: CV < 9.6 % |
| Recovery | Olanzapine/Desmethylolanzapine: 95 %; Quetiapine: 96 % |
| Specimen | Serum/Plasma |
| Sample Preparation | Extraction
|
| Run Time | 18 min |
| Injection Volume | 50 µl |
| Flow Rate | 0.6 ml/min |
| Column Temperature | ambient (~ 25 °C) |
| Gradient | isocratic |
| Wavelengths | 254 nm |
| Additional Info | Any isocratic HLPC system with UV-VIS detector is suitable. |
| Parameters | N-Desmethylolanzapine, Olanzapine, Quetiapine |
-
 Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
-
 HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC -
 Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
-
 Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Olanzapine/Desmethylolanzapine Plasma Calibration StandardOrder no.: 26003Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC -
 Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
Perazine, Quetiapine, Citalopram Plasma Calibration StandardOrder no.: 28007Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
-
 HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC
HPLC Column Olanzapine and Desmethylolanzapine in Serum/PlasmaOrder no.: 26100Olanzapine and Desmethylolanzapine in Serum/Plasma - HPLC