β-Thalassemia Testing - HPLC
Simultaneous quantifications of HbS and HbC within 5.5 minutes
HbA2 will be quantified in presence of HbE and HbD
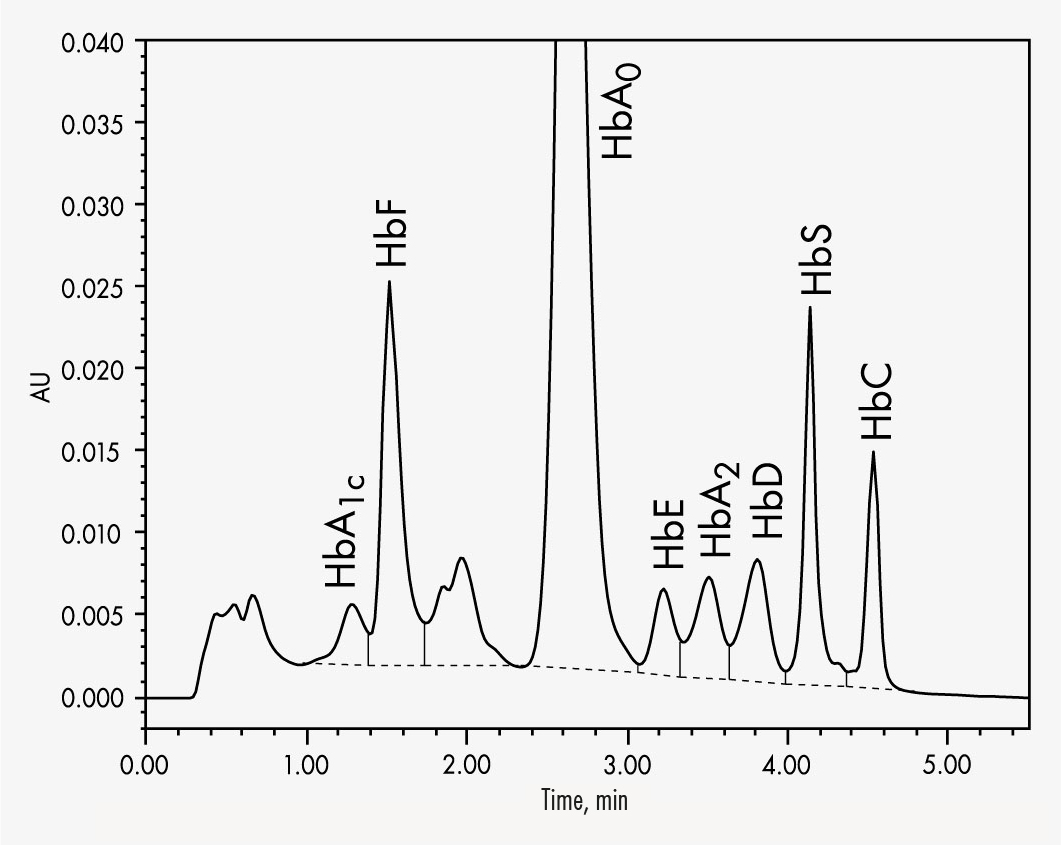


HbA1c
HbA2
HbC
HbF
HbS
Clinical relevance
Thalassaemias are caused by impaired synthesis of one or more globin chains of the haemoglobin (consisting of 4 polypeptide chains), resulting in much less oxygen being bound to the haemoglobin molecules and transported through the body. Depending on which polypeptide chains are affected, the thalassaemias are named α, β, γ or δ thalassaemia. It is the β chain that is most frequently affected, so that this disorder is called β-thalassaemia. β-thalassaemia major is a homozygous form in which both alleles are severely mutated so that β chain synthesis is stopped completely, whereas β-thalassaemia minor is a heterozygous form resulting in an approximately 20 % reduction of polypeptide synthesis. To compensate this reduction, more HbA2 and HbF are produced: in β-thalassaemia major it is more HbF, and in β-thalassaemia minor primarily HbA2.
Depending on severity, patients may suffer from enlargement of the liver and spleen, growth disorders, severe inner organ damages and bone deformities. Further mutations are known that modify the peptide chains in other ways, leading to abnormal haemoglobin variants. The most common and clinically relevant variants are HbS, HbC and HbE. HbD also occurs relatively often. Combinations of various thalassaemias with different genetically related haemoglobin variants are also possible.
Product advantages
- Simultaneous determination of HbS and HbC in less than 6 minutes
- Quantification of HbA2 without interferences from HbD and HbE variants
The Chromsystems assay for β-thalassaemia testing allows the reliable quantification of HbA2 and HbF. The column’s high separation capability allows the determination of the most common variants HbA1c, HbA2, HbC, HbF and HbS in less than 6 minutes. A column material specially designed for this specific separation process allows a stabilised separation of HbA2 from the other Hb-molecules even with aged columns. This helps to avoid misinterpretations and eliminate false quantifications and qualifications.
| Method of Analysis | HPLC |
|---|---|
| Number of Tests | 1000 |
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | HbA2: 2 % |
| Upper Limit of Quantification | HbA2: up to 75 % |
| Intraassay | CV ≤ 4.2 % |
| Interassay | CV ≤ 10 % |
| Recovery | 101 % |
| Specimen | Whole Blood |
| Sample Preparation |
|
| Run Time | < 5.5 min |
| Injection Volume | 10-20 µl |
| Flow Rate | 2.3 ml/min |
| Column Temperature | ambient (~ 25 °C) |
| Gradient | binary |
| Wavelengths | 415 nm |
| Additional Info | Any HPLC gradient system with UV/VIS detector is suitable. |
| Parameters | HbA1c, HbA2, HbC, HbF, HbS |
| Pressure | Maximum pressure: 140 bar |




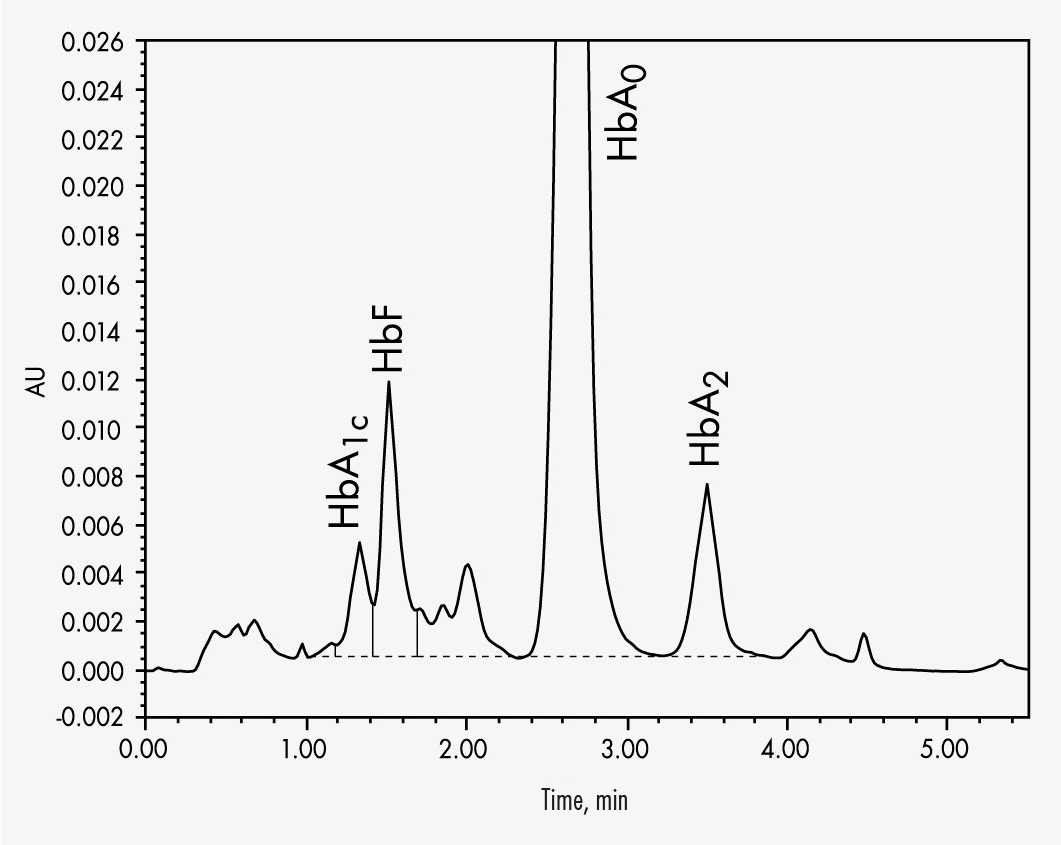
HbA1c
HbA2
HbC
HbF
HbS
Clinical relevance
Thalassaemias are caused by impaired synthesis of one or more globin chains of the haemoglobin (consisting of 4 polypeptide chains), resulting in much less oxygen being bound to the haemoglobin molecules and transported through the body. Depending on which polypeptide chains are affected, the thalassaemias are named α, β, γ or δ thalassaemia. It is the β chain that is most frequently affected, so that this disorder is called β-thalassaemia. β-thalassaemia major is a homozygous form in which both alleles are severely mutated so that β chain synthesis is stopped completely, whereas β-thalassaemia minor is a heterozygous form resulting in an approximately 20 % reduction of polypeptide synthesis. To compensate this reduction, more HbA2 and HbF are produced: in β-thalassaemia major it is more HbF, and in β-thalassaemia minor primarily HbA2.
Depending on severity, patients may suffer from enlargement of the liver and spleen, growth disorders, severe inner organ damages and bone deformities. Further mutations are known that modify the peptide chains in other ways, leading to abnormal haemoglobin variants. The most common and clinically relevant variants are HbS, HbC and HbE. HbD also occurs relatively often. Combinations of various thalassaemias with different genetically related haemoglobin variants are also possible.
Product advantages
- Simultaneous determination of HbS and HbC in less than 6 minutes
- Quantification of HbA2 without interferences from HbD and HbE variants
The Chromsystems assay for β-thalassaemia testing allows the reliable quantification of HbA2 and HbF. The column’s high separation capability allows the determination of the most common variants HbA1c, HbA2, HbC, HbF and HbS in less than 6 minutes. A column material specially designed for this specific separation process allows a stabilised separation of HbA2 from the other Hb-molecules even with aged columns. This helps to avoid misinterpretations and eliminate false quantifications and qualifications.
| Method of Analysis | HPLC |
|---|---|
| Number of Tests | 1000 |
| Please note | The freely available information on this website, in particular on the sample preparation, are not sufficient to work with our products. Please read instructions and warning notices on products and/or instruction manuals. |
| Lower Limit of Quantitation | HbA2: 2 % |
| Upper Limit of Quantification | HbA2: up to 75 % |
| Intraassay | CV ≤ 4.2 % |
| Interassay | CV ≤ 10 % |
| Recovery | 101 % |
| Specimen | Whole Blood |
| Sample Preparation |
|
| Run Time | < 5.5 min |
| Injection Volume | 10-20 µl |
| Flow Rate | 2.3 ml/min |
| Column Temperature | ambient (~ 25 °C) |
| Gradient | binary |
| Wavelengths | 415 nm |
| Additional Info | Any HPLC gradient system with UV/VIS detector is suitable. |
| Parameters | HbA1c, HbA2, HbC, HbF, HbS |
| Pressure | Maximum pressure: 140 bar |




