Introduction
Drugs of abuse are defined as substances that are consumed not for the treatment of disease, but for their mind-altering effects. This definition is not limited to illegal drugs; it also includes approved pharmaceuticals with abuse or habit-forming potential. Substances in this category include amphetamines, barbiturates, benzodiazepines, booster, cocaine, cannabinoids, opiates/opioids, Z-Drugs and other drugs of abuse,.
Here we describe a simple and quick adaptation of the MassTox® Drugs of Abuse Testing in urine kit (order no. 96000) to enable the analysis of 108 substances in venous whole blood. The changes only affect sample preparation, while the analytical system can remain completely unchanged.
The following substances can be determined:
11-Nor-9-carboxy-Δ9-THC
2C-B
2C-I
2-Oxo-3-hydroxy-LSD
3-Hydroxybromazepam
6-Monoacetylmorphine
7-Aminoclonazepam
7-Aminoflunitrazepam
7-Aminonitrazepam
Acetylcodeine
Allobarbital
Alprazolam
Amobarbital
Amphetamine
Barbital
BDB
Benzoylecgonine
Bromazepam
Brotizolam
Buprenorphine
Butalbital
Butylone
Cathinone
Chlordiazepoxide
Clobazam
Clonazepam
Cocaethylene
Cocaine
Codeine
Demoxepam
Desalkylflurazepam
Desmethylflunitrazepam
Diazepam
Dihydrocodeine
EDDP
Estazolam
Fentanyl
Flunitrazepam
Flurazepam
Gabapentin
Hexobarbital
Hydrocodone
Hydromorphone
Ketamine
Lorazepam
Lormetazepam
LSD
MBDB
MDA
MDEA
MDMA
MDPV
Meconin
Medazepam
Meperidine
Mephedrone
Mescaline
Methadone
Methamphetamine
Methaqualone
Methylone
Methylphenidate
Midazolam
Morphine
Naloxone
Naltrexone
Nitrazepam
Norbuprenorphine
Norclobazam
Norcocaine
Norcodeine
Nordiazepam
Norfentanyl
Norketamine
Normeperidine
Nortapentadol
Nortilidine
O-desmethyltramadol
Oxazepam
Oxycodone
Oxymorphone
apaverine
PCP
Pentobarbital
Phenobarbital
PMA
Prazepam
Pregabalin
Promethazine
Propoxyphene
Quetiapine
Ritalinic acid
Secbutabarbital
Secobarbital
Sufentanil
Tapentadol
Temazepam
Thebaine
Thiopental
Tilidine
Tramadol
Triazolam
Zaleplon
Zolpidem
Zopiclone
α-Hydroxyalprazolam
α-Hydroxymidazolam
α-Hydroxytriazolam
Material & Methods
Sample Preparation
Prior to sample preparation, the Internal Standard Solution is prepared by mixing the Internal Standard Mix (order no. 63004) and the Extraction Reagent (Order No. 63005) in an appropriate ratio of 1:9.
For sample preparation, 100 μl of Internal Standard Solution is pipetted into a 1.5 mL reaction vial. Subsequently, 20 μl of sample/calibrator/MassCheck® control is added and vortexed for 5 seconds, followed by centrifugation for 10 minutes at 15,000 g. The resulting supernatant is transferred into an autosampler vial.
LC-MS/MS
Injection volume of 1–20 µl is used. Chromatographic separation is performed on an Analytical Column (order no. 63100) at a column temperature of 40 °C. The analytes are separated under isocratic conditions at a flow rate of 0.4 ml/min using Mobile Phase (order no. 63001).
Electrospray ionization in negative ion mode is used. The multiple reaction monitoring (MRM) transitions used for quantification are listed in Table 1.
Quantification
A full calibration of the analysis system for each series of measurements is performed with 6PLUS1® multi level calibrator (order no. 63039). Calibration curves are constructed by calculating the analyte to internal standard (ISTD) peak area ratio on the y axis against calibrator concentrations on the x axis. Then a calibration curve is plotted for both PEth homolouges using linear regression and 1/x weighting.


Table 1: Multiple reaction monitoring transitions of Phosphatidylethanol 16:0/18:1 and 16:0/18:2, together with their respective internal standards
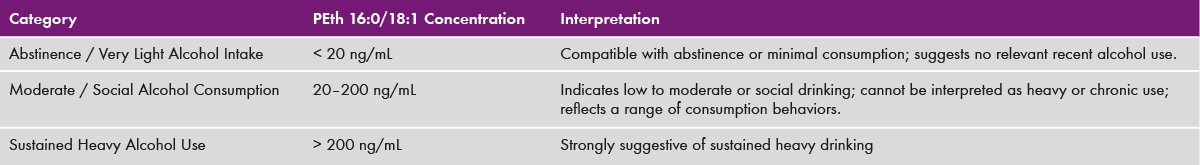

Table 2: Interpretation of Phosphatidylethanol 16:0/18:1 values
Results
The run time is 3.5 min (see chromatogram in fig. 1). The calibration range of 10-1000 ng/mL covers the forensic and clinical relevant concentrations (table 2).
Conclusion
This product insight describes a protocol for the determination of PEth 16:0/18:1 and PEth 16:0/18:2 in whole blood using Chromsystems components. Suitable products for implementation in your laboratory will soon be available from Chromsystems.
Outlook
Further research will focus on developing a reliable protocol for automated sample preparation using 96-well plates and liquid handling systems.
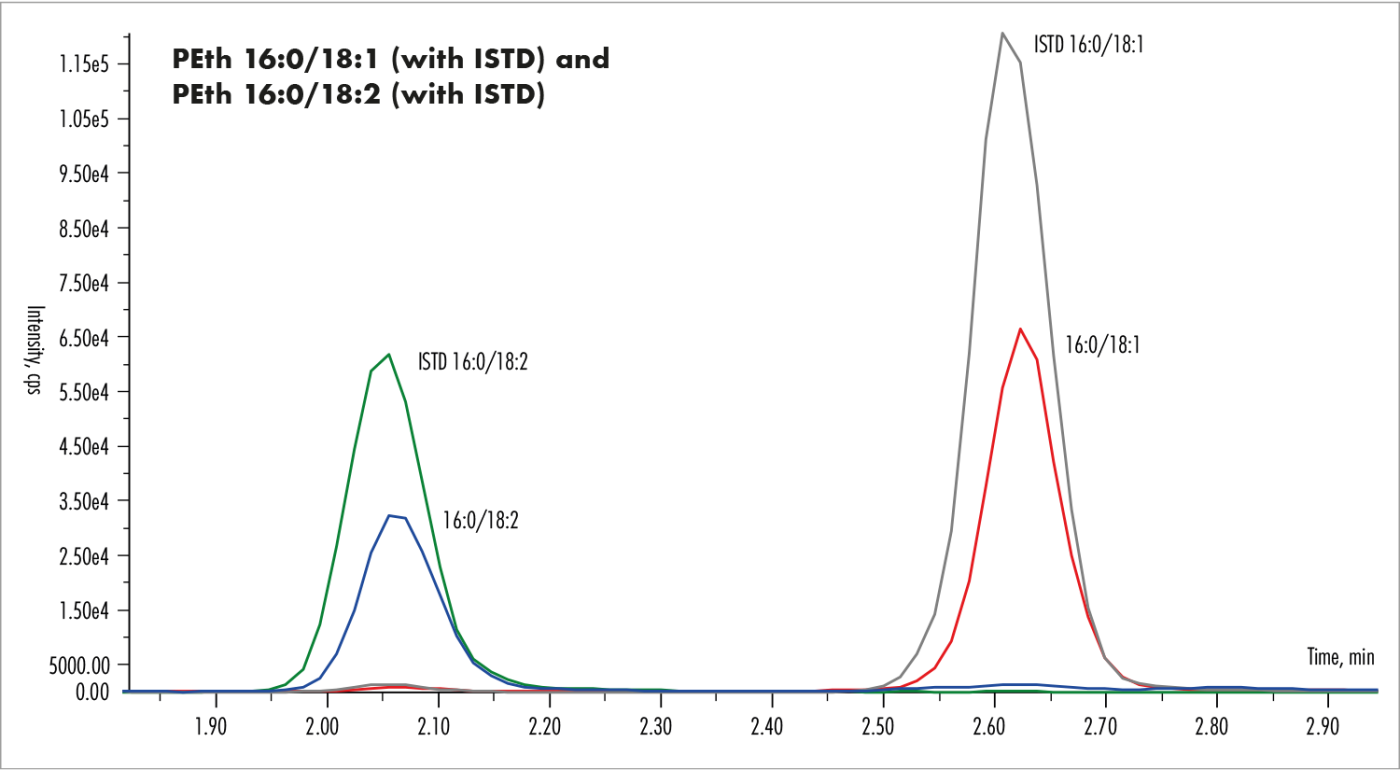

Fig. 1: Chromatogram of phosphatidylethanol 16:0/18:1 and 16:0/18:2 at a concentration of 300 ng/mL each, with their respective internal standards

Fig. 2: Chemical structure of Phosphatidylethanol 16:0/18:1 and 16:0/18:2
Ordering Information (To order the products or for a quote, please contact us here)
| Product | Order no. |
| Mobile Phase | 63001 |
| Rinsing Solution | 63009 |
| Internal Standard Mix | 63004 |
| Extraction Reagent | 63005 |
| Tuning Mix, Analytes and Internal Standards | 63015 |
| Analytical Column | 63100 |
| Product | Order no. |
| 6PLUS1® Multilevel Whole Blood Calibrator Set MassChrom® Phosphatidylethanol (PEth) |
63039 |
| MassCheck® Phosphatidylethanol (PEth) Whole Blood Control Set Level I |
6310 |
| MassCheck® Phosphatidylethanol (PEth) Whole Blood Control Set Level II |
6320 |
Last Update March 2026
